Cookie-Einstellungen
Diese Website benutzt Cookies, die für den technischen Betrieb der Website erforderlich sind und stets gesetzt werden. Andere Cookies, die den Komfort bei Benutzung dieser Website erhöhen, der Direktwerbung dienen oder die Interaktion mit anderen Websites und sozialen Netzwerken vereinfachen sollen, werden nur mit Ihrer Zustimmung gesetzt.
Konfiguration
Technisch erforderlich
Diese Cookies sind für die Grundfunktionen des Shops notwendig.
"Alle Cookies ablehnen" Cookie
"Alle Cookies annehmen" Cookie
Ausgewählter Shop
CSRF-Token
Cookie-Einstellungen
FACT-Finder Tracking
Individuelle Preise
Kundenspezifisches Caching
Session
Währungswechsel
Komfortfunktionen
Diese Cookies werden genutzt um das Einkaufserlebnis noch ansprechender zu gestalten, beispielsweise für die Wiedererkennung des Besuchers.
Facebook-Seite in der rechten Blog - Sidebar anzeigen
Merkzettel
Statistik & Tracking
Endgeräteerkennung
Kauf- und Surfverhalten mit Google Tag Manager
Partnerprogramm
Bei Fragen nutzen Sie gerne unser Kontaktformular.
Bestellen Sie auch per E-Mail: info@biomol.com
Größere Menge gewünscht? Bulk-Anfrage
Bestellen Sie auch per E-Mail: info@biomol.com
Größere Menge gewünscht? Bulk-Anfrage
Organism: Homo sapiens (Human). Source: E.coli. Expression Region: 151-250aa. Protein Length:... mehr
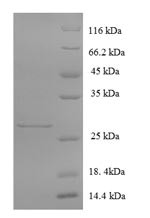
Produktinformationen "Glutamate receptor 3 (GRIA3), partial, human, recombinant"
Organism: Homo sapiens (Human). Source: E.coli. Expression Region: 151-250aa. Protein Length: Partial. Tag Info: N-terminal 6xHis-SUMO-tagged. Target Protein Sequence: SLLGHYKWEK FVYLYDTERG FSILQAIMEA AVQNNWQVTA RSVGNIKDVQ EFRRIIEEMD RRQEKRYLID CEVERINTIL EQVVILGKHS RGYHYMLANL. Purity: Greater than 90% as determined by SDS-PAGE. Endotoxin: Not test. Biological Activity: n/a. Form: Liquid or Lyophilized powder. Buffer: If the delivery form is liquid, the default storage buffer is Tris/PBS-based buffer, 5%-50% glycerol. If the delivery form is lyophilized powder, the buffer before lyophilization is Tris/PBS-based buffer, 6% Trehalose, pH 8.0. Reconstitution: We recommend that this vial be briefly centrifuged prior to opening to bring the contents to the bottom. Please reconstitute protein in deionized sterile water to a concentration of 0.1-1.0 mg/mL.We recommend to add 5-50% of glycerol (final concentration) and aliquot for long-term storage at -20 °C/-80 °C. Our default final concentration of glycerol is 50%. Customers could use it as reference. Storage: The shelf life is related to many factors, storage state, buffer ingredients, storage temperature and the stability of the protein itself. Generally, the shelf life of liquid form is 6 months at -20 °C/-80 °C. The shelf life of lyophilized form is 12 months at -20 °C/-80 °C. Notes: Repeated freezing and thawing is not recommended. Store working aliquots at 4 °C for up to one week. Relevance: Receptor for glutamate that functions as ligand-gated ion channel in the central nervous syst and plays an important role in excitatory synaptic transmission. L-glutamate acts as an excitatory neurotransmitter at many synapses in the central nervous syst. Binding of the excitatory neurotransmitter L-glutamate induces a conformation change, leading to the opening of the cation channel, and thereby converts the chical signal to an electrical impulse. The receptor then desensitizes rapidly and enters a transient inactive state, characterized by the presence of bound agonist. In the presence of CACNG4 or CACNG7 or CACNG8, shows resensitization which is characterized by a delayed accumulation of current flux upon continued application of glutamate. Reference: The DNA sequence of the human X chromosome.Ross M.T., Grafham D.V., Coffey A.J., Scherer S., McLay K., Muzny D., Platzer M., Howell G.R., Burrows C., Bird C.P., Frankish A., Lovell F.L., Howe K.L., Ashurst J.L., Fulton R.S., Sudbrak R., Wen G., Jones M.C. , Hurles M.E., Andrews T.D., Scott C.E., Searle S., Ramser J., Whittaker A., Deadman R., Carter N.P., Hunt S.E., Chen R., Cree A., Gunaratne P., Havlak P., Hodgson A., Metzker M.L., Richards S., Scott G., Steffen D., Sodergren E., Wheeler D.A., Worley K.C., Ainscough R., Ambrose K.D., Ansari-Lari M.A., Aradhya S., Ashwell R.I., Babbage A.K., Bagguley C.L., Ballabio A., Banerjee R., Barker G.E., Barlow K.F., Barrett I.P., Bates K.N., Beare D.M., Beasley H., Beasley O., Beck A., Bethel G., Blechschmidt K., Brady N., Bray-Allen S., Bridgeman A.M., Brown A.J., Brown M.J., Bonnin D., Bruford E.A., Buhay C., Burch P., Burford D., Burgess J., Burrill W., Burton J., Bye J.M., Carder C., Carrel L., Chako J., Chapman J.C., Chavez D., Chen E., Chen G., Chen Y., Chen Z., Chinault C., Ciccodicola A., Clark S.Y., Clarke G., Clee C.M., Clegg S., Clerc-Blankenburg K., Clifford K., Cobley V., Cole C.G., Conquer J.S., Corby N., Connor R.E., David R., Davies J., Davis C., Davis J., Delgado O., Deshazo D., Dhami P., Ding Y., Dinh H., Dodsworth S., Draper H., Dugan-Rocha S., Dunham A., Dunn M., Durbin K.J., Dutta I., Eades T., Ellwood M., Emery-Cohen A., Errington H., Evans K.L., Faulkner L., Francis F., Frankland J., Fraser A.E., Galgoczy P., Gilbert J., Gill R., Gloeckner G., Gregory S.G., Gribble S., Griffiths C., Grocock R., Gu Y., Gwilliam R., Hamilton C., Hart E.A., Hawes A., Heath P.D., Heitmann K., Hennig S., Hernandez J., Hinzmann B., Ho S., Hoffs M., Howden P.J., Huckle E.J., Hume J., Hunt P.J., Hunt A.R., Isherwood J., Jacob L., Johnson D., Jones S., de Jong P.J., Joseph S.S., Keenan S., Kelly S., Kershaw J.K., Khan Z., Kioschis P., Klages S., Knights A.J., Kosiura A., Kovar-Smith C., Laird G.K., Langford C., Lawlor S., Leversha M., Lewis L., Liu W., Lloyd C., Lloyd D.M., Loulseged H., Loveland J.E., Lovell J.D., Lozado R., Lu J., Lyne R., Ma J., Maheshwari M., Matthews L.H., McDowall J., McLaren S., McMurray A., Meidl P., Meitinger T., Milne S., Miner G., Mistry S.L., Morgan M., Morris S., Mueller I., Mullikin J.C., Nguyen N., Nordsiek G., Nyakatura G., O'dell C.N., Okwuonu G., Palmer S., Pandian R., Parker D., Parrish J., Pasternak S., Patel D., Pearce A.V., Pearson D.M., Pelan S.E., Perez L., Porter K.M., Ramsey Y., Reichwald K., Rhodes S., Ridler K.A., Schlessinger D., Schueler M.G., Sehra H.K., Shaw-Smith C., Shen H., Sheridan E.M., Shownkeen R., Skuce C.D., Smith M.L., Sotheran E.C., Steingruber H.E., Steward C.A., Storey R., Swann R.M., Swarbreck D., Tabor P.E., Taudien S., Taylor T., Teague B., Thomas K., Thorpe A., Timms K., Tracey A., Trevanion S., Tromans A.C., d'Urso M., Verduzco D., Villasana D., Waldron L., Wall M., Wang Q., Warren J., Warry G.L., Wei X., West A., Whitehead S.L., Whiteley M.N., Wilkinson J.E., Willey D.L., Williams G., Williams L., Williamson A., Williamson H., Wilming L., Woodmansey R.L., Wray P.W., Yen J., Zhang J., Zhou J., Zoghbi H., Zorilla S., Buck D., Reinhardt R., Poustka A., Rosenthal A., Lehrach H., Meindl A., Minx P.J., Hillier L.W., Willard H.F., Wilson R.K., Waterston R.H., Rice C.M., Vaudin M., Coulson A., Nelson D.L., Weinstock G., Sulston J.E., Durbin R.M., Hubbard T., Gibbs R.A., Beck S., Rogers J., Bentley D.R.Nature 434:325-337(2005). Function: Receptor for glutamate that functions as ligand-gated ion channel in the central nervous system and plays an important role in excitatory synaptic transmission. L-glutamate acts as an excitatory neurotransmitter at many synapses in the central nervous system. Binding of the excitatory neurotransmitter L-glutamate induces a conformation change, leading to the opening of the cation channel, and thereby converts the chemical signal to an electrical impulse. The receptor then desensitizes rapidly and enters a transient inactive state, characterized by the presence of bound agonist. In the presence of CACNG4 or CACNG7 or CACNG8, shows resensitization which is characterized by a delayed accumulation of current flux upon continued application of glutamate.
| Schlagworte: | GluA3, GLUR3, GRIA3, GluR-3, GluR-C, GluR-K3, Glutamate receptor 3, AMPA-selective glutamate receptor 3, Glutamate receptor ionotropic, AMPA 3, Recombinant Human Glutamate receptor 3 (GRIA3), partial |
| Hersteller: | Cusabio |
| Hersteller-Nr: | EP009900HU |
Eigenschaften
| Anwendung: | Activity not tested |
| Konjugat: | No |
| Wirt: | E.coli |
| Spezies-Reaktivität: | human |
| MW: | 28 kD |
| Reinheit: | >90% (SDS-PAGE) |
Datenbank Information
| KEGG ID : | K05199 | Passende Produkte |
| UniProt ID : | P42263 | Passende Produkte |
| Gene ID : | GeneID 2892 | Passende Produkte |
Handhabung & Sicherheit
| Lagerung: | -20°C |
| Versand: | +4°C (International: +4°C) |
Achtung
Nur für Forschungszwecke und Laboruntersuchungen: Nicht für die Anwendung im oder am Menschen!
Nur für Forschungszwecke und Laboruntersuchungen: Nicht für die Anwendung im oder am Menschen!
Hier kriegen Sie ein Zertifikat
Loggen Sie sich ein oder registrieren Sie sich, um Analysenzertifikate anzufordern.
Bewertungen lesen, schreiben und diskutieren... mehr
Kundenbewertungen für "Glutamate receptor 3 (GRIA3), partial, human, recombinant"
Bewertung schreiben
Loggen Sie sich ein oder registrieren Sie sich, um eine Produktbewertung abzugeben.
Zuletzt angesehen

